Boron trifluoride (etherate) Method of Analysis
Boron trifluoride Etherate
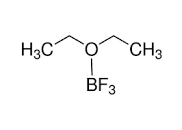
Following is the structure of Boron trifluoride etherate:
(Structure drawn in ACD/ChemSketch)
From the structure, it is a Lewis acid. Hence it can be easily quantified using acid-base titration. Its other properties are:
- Molecular formula: BF3O(C2H5)2
- Molecular weight: 141.93g/mol
- Appearance: Colourless liquid
Reagents and chemicals required for analysis:
- 0.1N Sodium hydroxide solution (of known normality)
- Mannitol
- Phenolphthalein indicator (Dissolve 1.0 gram of Phenolphthalein in 1000 ml of alcohol)
Boron trifluoride Etherate Assay procedure:
Weigh about 200 mg of sample and transfer in a 200 ml stopper flask containing 50 ml of water. Close and allow to stand for 5 minutes. Add 2 to 3 drops of Phenolphthalein indicator. and titrate with 0.1N sodium hydroxide solution. Add 200 to 300mg of Mannitol and continue the titration with 0.1N Sodium hydroxide. If the colour disappears then repeat the titration until the colour remains stable. Each ml of 0.1N Sodium hydroxide corresponds to 2.2606 mg of BF3.
Calculation
% Assay of BF3 = {(Vs-Vb)×F×0.0022606X100s}⁄W
Where: Vs is the Volume of titrant in ml used for the sample, Vb is the Volume of titrant used for blan, F is the factor of 0.1N Sodium hydroxide and W is the Sample weight in gram
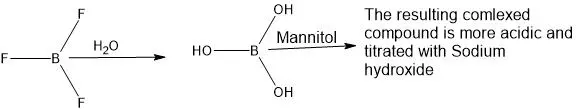
Why Mannitol is used in the assay titration of trifluoride etherate?
In the presence of water BF3 is hydrolyzed to Boric acid. The Boric acid is a weak monobasic acid which cannot be directly titrated with sodium hydroxide using phenolphthalein as an indicator. To enhance the acidic strength of the Boric acid, it is complexed with Mannitol and then titrated with Sodium hydroxide.



Dear sir,
I can not understand below listed queries.
What is principle of this titration.
Which is role of mannitol in this titration
Your answer has been included in the post. Please check.
Thank you sir,
understand principle of this titration
very good uses of Lewis acid principle
If we boil the solution of BF3 and water (Reflux) , would’nt HF be formed? The same can be titrated??